Tilapia
Contents
- 1 Feeding
- 2 Protein
- 3 Fat
- 4 Carbohydrates
- 5 Chito-oligosaccharides
- 6 Vitamins & Minerals
- 7 Supplements
- 8 Dissolved oxygen
- 9 Salinity
- 10 Alkalinity & pH
- 11 Water flow
- 12 Stocking density
- 13 Growth
- 14 Broodstock
- 15 Incubation
- 16 Strains
- 17 Masculinization
- 18 Feminization
- 19 Genetic Masculinization
- 20 Parasites
- 21 Author
Feeding




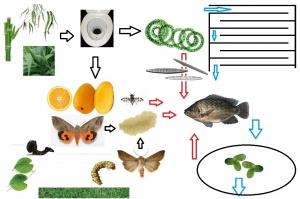
Tilapia may survive on algae or duckweed alone, but combined feeding results in higher growth rates. Feeding on algae. feeding most intensively occurs between 12.00 and 18.00, and in this time span the fish may consume over 3% of their bodyweight.[8] Feed efficiency is highest at 2% BW.[9] Tilapia may also feed on insects / larvae [10], such as water fleas (Ceriodaphnia < 1mm long) [11] (1> Daphnia <5 mm long; which cannot easily avoid predation) [12] (or Daphniamenucoensis, which are halophylic), Chaoborus midge larva (< 2cm) and Chironomus midges.[13] Superior growth is achieved by partial replacement with feed from animal origin, such as commercial fishfeed or insects (eg fruit flies) and worms. Water transparency contributes to the inclusion of insects in Nile tilapia diets.[14] Protein and energy of the animal-based foodstuffs are more available to Oreochromis niloticus than that of the plant-based foodstuffs.[15] Tilapia eat Spirulina platensis at a rate of 29 g dry weight / kg bodyweight, resulting in a daily growth of 14 g / kg bodyweight (almost 50% conversion rate).
In outdoor tanks fecundity and final mean weight is higher in tilapia fed 3% bw compared to lower feeding rates.[16] In adult tilapia, the optimal interval between feedings is 4 to 5 hours. At shorter intervals, eaten food will surpass the already filled stomach, and become waste. Fry may be fed 8 to 10 times a day.[17] Energy retention is highest in juvenile Tilapia (av. 34 g.) fed 3 times daily. Protein retention is highest in fish fed 5 times daily.[18] Tilapia may fast up to a week with compensatory enhanced growth after refeeding.[19]
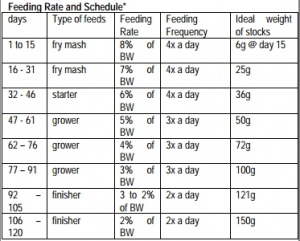
Table 1 (on the left) can be used by fish farmers to adjust feeding rate and frequency after determination of the hatchery constant by sampling and weighing the fish. The hatchery constant will depend on water quality, stocking density etc.[20] Table 2 (on the left) is a schedule for the administration of commercial fish feed. Larger feed particle sizes are associated with lower fish growth rates.[21]
Protein
Protein requirement for maximum performance of tilapia larva is 35 -> 50%, decreasing with increasing fish size.[22][23][24] In tilapia fry the feed conversion ratio and the highest protein growth rate are highest with a 45% crude protein diet, but the protein efficiency ratio and protein productive value was highest with the 25% crude protein diet. Fingerlings and advanced juveniles showed optimum growth performance with the 35% crude protein diet.[25] In young tilapia, out of artificial diets with dietary protein levels of 20%, 30%, 40% and 50%, best growth and food conversion efficiency was obtained with 40% dietary protein.[26] Maximum food conversion efficiency and growth of young tilapia was obtained using the diet containing 30% protein (compared to 20%, 40% and 50% protein), decreasing with increasing protein levels.[27] Juvenile tilapia need 30-40% protein, while adult tilapia need 20-30% dietary protein for optimum performance. Tilapia broodstock need 35-40% protein for optimum reproduction, spawning efficiency, and larval growth and survival.[28] [29][30]. In tilapia average 62 g. weight and 18 cm long, there was no significant increase in growth rate with increasing dietary protein levels (from 25% to 30%).[31] Excess protein results in unionized ammonia in the environment.[32]
Duckweeds grown in enriched water may contain 30 to 40% protein.[33] Wolffia arrhiza (dry weight) duckweed may contain up to 40% protein.[34] Lemna paucicostata dry mass protein contents may range from 26% to 45%.[35] Lemna minor grown in swine lagoon wastewater contained 32% protein.[36]
Green algae (Chlorella vulgaris, Scenedesmus obliquus) and blue-green algae (Anacystis nidulans, Microcystis aeruginosa, Oscillatoria rubescens, Spirulina platensis) may accumalate protein from 8 to 54% as available nitrogen increases. [37] Spirulina platensis contains 50 to 65% protein (10% is non-protein nitrogen)[38], or even up to 71.9% protein.[39] At high salinities, protein levels in Spirulina decrease while carbohydrates increase as an adaptation for efficient osmoregulation. [40]
Insect true protein contents may be reasonably estimated via crude protein (nitrogen x 6.25).[41] Crude protein content of feeder insects (housefly, fly larvae, cockroach nymphs, tebo worms) may range from 15.5 to 19.7% [42] Total crude protein in dry matter is 32.5% in earth worms and 64.4% in adult crickets (water content >50%) Protein contents of Armyworm (Spodoptera exempta) moths are similar in gregarious and solitary moths.[43]
Black soldier fly larvae (Hermetia illucens) may contain up to 42% of protein.
Silkworm (Bombyx mori) pupae are a rich source of high quality protein.[44] In silkworm larvae, two storage proteins may account for 60% of total fat body protein (80% of the soluble protein) in females, compared to only 20% of the total fat body protein in males.[45]
Mealworms (Tenebrio molitor) from commercial source contained 22.3% protein.[46] Mealworms (Tenebrio molitor) fed fermented grain, leaves and vegetable wastes reached half the weight of conventionally reared larvae but contained 76% protein on dry weight basis.[47] Mealworms contain 53% (Tenebrio molitor) and 45% (Zophobas morio; 'Superworm') protein in dry matter.[48] (Egg production in Tenebrio molitor is 640 eggs / female / year, and 1500 eggs / female / year in Zophobas morio) Yellow mealworms (Tenebrio molitor; 4.8-182.7 mg) fed on wheat flour and brewers yeast contained 24.3-27.6% protein.[49] A 40% protein fish meal diet with up to 40% replacement of fish meal with mealworm (Tenebrio molitor) meal does not affect growth of catfish. Catfish fed diets with up to 80% replacement of fish meal with the worm meal still displayed good growth and feed utilization efficiency. [50] Cuticle larval / pupal mealworm proteins are more water-soluble at low temperatures (4 to 10°C) and become more hydrophobic at 25°C.[51] These cuticle proteins (as in the locust) lack acidic amino acid residues, methionine, cysteine and tryptophan.[52]
The digestibility coefficients for protein in Oreochromis niloticus are: soybean meal (91%), fish meal (86%), ground corn (83%), wheat middlings (75%), poultry-offal meal (74%), and brewers grain (63%).[53]
Protease activity in Tilapia is relatively low, in comparison to Grass carp (Ctenopharyngododn idellus), Common carp (Cyprinus carpiol), silver carp (Hypophthaimichthys molitrix) and bighead carp (Aristichys nobilis Richardson).[54] Alkaline protease activity in Oreochromis niloticus is significantly inhibited by the ingestion of soybean meal, corn gluten meal and wheat bran, in comparison to the sensitivity to protease inhibitors in seabream and sole.[55]
Fat






Tilapia do well on fat versus carbohydrates as a source of energy.[56] Excess dietary lipids, however, are readily stored in the carcass and the viscera, but are not utilized for improving growth or food utilization efficiency.[57]
Larvae of mealworms, crickets, waxworms and fruit flies (Drosophila melanogaster) on average have 30% of dry matter higher fat content than adult species.[58] Newly emerged Armyworm moths may (still) contain high levels of (glyceride [59]) lipid (largely in the abdominal), the quantity depends on the larval feeding conditions. Both the Armyworm larvae and young adult moths have the capacity to accumulate lipid reserves in excess. Big female moths contain relatively much fat. The moths are able to supplement their lipid reserves following carbohydrate uptake.[60] Gregarious larva have 2.5 to 6.1 higher abdominal glyceride contents than solitaria phase moths.[61] The stored fat may serve as fuel for long migratory flights.[62] In feeder insects (house fly, fly larvae, cockroach nymphs, tebo worms), crude fat may range from 1.9% to 29.4%.[63] Yellow mealworms (4.8-182.7 mg) fed on wheat flour and brewers yeast contained 12.0-12.5% fat.[64] Mealworms (Tenebrio molitor) from commercial source contained 15% fat.[65] Mealworms (Tenebrio molitor) fed fermented grain, leaves and vegetable wastes reached half the weight of conventionally reared larvae and contained 6.4% fat on dry weight basis.[66]
Energy content for silkworms is 674 kcal/kg and 2741 kcal/kg for waxworms.[67]
In regard to gross energy, the digestibilities in Oreochromis niloticus are: animal oil (93%), fish meal (80%), ground corn (76%), poultry-offal meal (59%), wheat middlings (58%), soybean meal (56%), and brewers grain (30%).[68] Replacing sunflower oil (69% omega-6; 0% omega-3) with perilla oil (50 to 60% ALA/omega-3) changed fatty acid profiles in Nile tilapia, which stabilized after 20 days under the new diet. Total omega-3 in muscle tissue fat increased from 6.4% to 18.2% (almost 3-fold), particularly due to a 9.4 fold increase in ALA levels.[69] Palm oil may replace soybean oil in feeds for Oreochromis niloticus fingerlings without any negative effect on growth or body composition.[70]
Spirulina may contain 7% [71] to 14% fat (of dry mass)[72]. At low nitrogen (N) levels, green algae (Chlorella vulgaris and Scenedesmus obliquus) contain 45% lipids of biomass, containing mainly 16:0 and 18:1 fatty acids. At high N levels, total lipids dropped to 20% of the dry weight, with elevated levels of polyunsaturated C16 and C18 fatty acids [[73] during the initial stages of growth. Towards the end of growth the main lipids contained mainly saturated fatty acids (mostly 18:1 and 16:0).[74] In blue-green algae (Anacystis nidulans, Microcystis aeruginosa, Oscillatoria rubescens, Spirulina platensis) lipid composition was reported not to be affected by N concentrations [75], but by sodium-nitrate as well as by temperature.[76] The fatty acids 18:3n−3, 18:2n−6, 18:0, 18:1 and 16:0 dominate in microalgae (eg Chlorella vulgaris, Scenedesmus abundans, Monoraphidium minitum), but are highest in Chlorella vulgaris. This difference is not reflected in their predator, Brachionus calyciflorus, and in Tilapia feeding on Brachionus calyciflorus. The Tilapia zillii larvae contained relatively much 22:6n−3, which is indicative for the capacity to elongate and desaturate dietary linoleic and linolenic acid.[77]
Carbohydrates
Tilapia is relatively well equipped (due to alpha-Amylases[78] produced by Aeromonas, Bacteroidaceae and Clostridium strains[79]) for utilizing carbohydrates.[80] Tilapia can utilize up to 46% dietary starch without growth retardation.[81] Tilapias do better on starch than on glucose (even when the glucose diet is supplemented with chromic oxide) [82] Starch consists of amylose and amylopectin. Amylopectin is more susceptible to enzymatic cleavage. A high-dietary amylose-amylopectin ratio (> 0.24; as in corn starch) decreases starch digestibility and postprandial peak blood glucose and triglycerides, and inhibits feed efficiency and growth in tilapia.[83] Amylopectin accounts for about 75% of the starch weight in reserve starch (in storage organs) [84] and typically more than 90% in transitory starch (in photosynthetic organs)[85]. Spirodela polyrrhiza amylose content was 21%[86], compared to 25% in potatoes[87], 7-44% in barley[88] and 25–28 in wheat.[89] Feeds with smaller granule size have higher starch digestibility than those with bigger granules. Spirodela polyrrhiza granules range from 1 to 8 μ [90], compared to 2-55 μ in wheat starch [91], 7 to 22 µ in corn starch and 8 to 110 µ in potatoes [92].
Oreochromis niloticus weight gain, feed conversion, specific growth rate and protein efficiency ratio were improved with diets containing date (15%, 30% and 45%) as compared with the starch diet (0% date diet). The diet containing 30% date was superior to all other test diets in terms of all the above growth parameters, while body fat was reduced.[93]
In tilapia, polysaccharides have antibacterial properties, increasing resistance to infections.[94] Spirulina platensis contains 13.6% carbohydrate, principally glucose along with rhamnose, mannose, xylose, galactose and two unusual sugars (2-O-methyl-L-rhamnose and 3-O-methyl-L-rhamnose).[95] Spirulina platensis contains various polysaccharides [96], including starch.[97] The amount of starch is low due to the high activity of α and β amylase in Spirulina.[98] In Spirulina platensis, the polysaccharide SP-4 consists of L-rhamnose, L-fucose, L-arabinose, D-glucose, D-glucoseamine and D-galacturonic acid. It enhances proliferation of lymphocytes.[99] The polysaccharide PSP in Spirulina platensis is composed of L-fucose, D-mannose, D-galactose, D-glucose and glucuronic acid.[100] Another polysaccharide in Spirulina platensis contains glucose, principally, with small amounts of sulphate; a glucan backbone with glucosyl sidechains.[101] Some Spirulina polysaccharides have antiviral properties.[102][103] Spirulina platensis also secretes polysaccharides.[104] The cell wall of Spirulina platensis lacks cellulose (and is therefore readily digested).[105]
The cell walls of green algae (Chlorophyta) contain cellulose (and pectose, the outer layer) and carbohydrates are stored as starch. Mealworms (Tenebrio molitor) from commercial source contained 3.6% carbohydrates.[106]
Chito-oligosaccharides
In tilapia stomach, intestine, and serum, chitinases are present. [107] Chitinases are enzymes required to enzymatically decompose chitin. Chitin is an amino polysaccharide; a long chain of many monosaccharide (N-acetylglucosamine) units. A chito-oligosaccharide is a short chain of just a few linked units of N-acetylglucosamine. Chitin may be split into chito-oligosaccharides. Chitin plus other composite materials (eg glycoproteins, calcium carbonate) forms the exoskeletons of insects and the flexible body wall of larva / caterpillars (eg silkworms[108] and fruit flies[109]). Apparent digestibility coefficient of chitin ingested by Oreochromis niloticus from crayfish exoskeleton meal was 69.3%.[110] The peritrophic matrix of the tobacco hornworm (Manduca sexta) may contain 40% chitin.[111] (Peritrophic matrices usually contain 3 to 13% chitin[112]) Wild-caught earthworms may contain 51% chitin of dry matter.[113] Crustaceans may contain 12 to 20% chitin.[114] Arthropod species (eg crustaceans, arachnids) with a chitin content of over 50% (of total weight) are eliminated unchanged by Tilapia zillii [115] Insects may contain 0.3 to 5% chitin. (1.2 to 14% of dry matter)[116] Chitin (and cellulose, murein, protein, silica and calcium carbonate [117]) is also an ingredient of cell walls in algae.[118][119] Dietary intake of chito-oligosaccharides can improve intestinal health, and improve tilapia resistance to infection.[120] Dietary intake of chito-oligosaccharides at up to 4 g/kg may considerably improve growth, survival and immune response.[121] Dietary intake of chito-oligosaccharides at 3 to 5 g/kg for juvenile GIFT tilapia may increase growth performance, nonspecific immunity and promote blood lipid metabolism.[122]
Vitamins & Minerals
Oxalic acid is an anti-nutrient, reducing nutrient conversion in tilapia. High calcium available to duckweeds results in higher duckweed oxalic acid contents.[123] Lower duckweed-calcium may be compensated for by calcium-carbonate in the water, as freshwater fish take up Ca2+ predominantly through the gills [124], or by higher calcium in worms fed to tilapia. In mealworms supplemented with Ca from CaCO3 (4 to 12%), the Ca content of the mealworms increased linearly with the Ca content of the substrate during the first 24 hours and decreased after over one week, especially at the higher levels of Ca supplementation. The Ca in these mealworms was 76% as bioavailable. [125] Ca concentrations of silkworms also increased in a linear fashion with increasing levels of dietary Ca. Gut-loading diets for mealworms should be supplemented to contain 9% calcium, iron (51 mg/kg) and manganese (31 mg/kg). Gut-loading diets for silkworms should be supplemented to contain 23 g calcium per kg feed.[126]
Insects (mealworms, waxworms, crickets, fruitflies) on average have low calcium concentrations (0.11%), but sufficient concentrations of Cu, Fe, Mg, P, and Zn to meet known requirements of domestic birds and mammals. Supermealworms (Zophobas morio) and waxworms contain deficient levels of manganese. Earthworms meet all dietary mineral requirements. [127]
Intestinal microorganisms in Tilapia nilotica produce at least 11.2 ng of vitamin B12 per g of body weight per day. In 16-weeks without Dietary B12, fish weight increased 8-fold (normal growth), and liver-stored B12 did not decrease.[128] Magnesium absorption from the gastrointestinal tract may be highly efficient in Oreochromis mossambicus.[129]
Supplements


- Saponin-rich plants such as Quillaja saponaria (Soap bark tree), yucca and Sapindus saponaria (wingleaf soapberry, western soapberry, jaboncillo) may increase growth, increase the nce male to female ratio in tilapia [130] and increase serum cholesterol in males.[131] Gypsophila paniculata (baby's breath) and Quillaja saponaria (both high in saponins) may induce release of LH (Luteinizing hormone)[132] Supplementing Tilapia fry with 700 mg/kg extracted Quillaja saponin extract resulted in significantly increased growth, higher number of males, higher LH release and higher serum cholesterol.[133] Saponin supplementation diminishes egg production by females.[134] Intestinal magnesium uptake in tilapia is stimulated by chloride and inhibited by saponins (which may induce intravesicular magnesium accumulation.[135] In tilapia, saponins selectively permeabilize the plasma membrane.[136]
- Allspice Pimenta dioica (10 g/kg fish) acts as a growth promoter to improve feed utilization and weight gain in Mozambique Tilapia fry and acts an antimicrobial agent to enhance disease resistance during first feeding of fry.[137] Allspice (aka Jamaica pepper, myrtle pepper, pimenta, pimento, English pepper, newspice) is dried unripe berries of Pimenta dioica.
- Honey bee pollen (2.5% (w/v)) increases growth performance parameters, feed efficiency ratio and immunological, hematological and biochemical parameters in young Nile tilapia.[138]
- A supplemental mixture (at 0.5 to 2% w/w) composed of astragalus, angelica, hawthorn, Licorice root and honeysuckle may increase lysozyme, superoxide dismutase and peroxidase activity, decrease malondialdehyde levels and reduce mortality in Aeromonas hydrophila-challenged tilapia.[139] 20 days administration of 1% Astragalus root (Radix astragalin seu Hedysari) plus Angelica Root (Angelicae Sinensis) at a ratio of 5:1 (w/w) increased body weight of carp relative to controls.[140] Another study found no effect on growth by an mixture of Astragalus membranaceus, orange peel, hawthorn, pilose asiabell root indigowoad root, taraxacum and malt.[141] Astragalus (aka milkvetch, locoweed) belongs to the legume family Fabaceae. Dietary Astragalus polysaccharides supplementation may improve growth performance and immune parameters of tilapia.[142] Calycosin is a major isoflavonoid extracted from astragalus. It has shown to have proangiogenic effects.[143] Two isomeres extracted from Astragalus membranceus may stimulate proliferation in human fetal lung diploid fibroblast cells.[144]
- Survival in Streptococcus agalactiae challenged tilapia is increased by inclusion of 0.1% Sophora flavescens (root extract) in the diet.[145] Sophora flavescens (member of the Fabaceae family) roots contain quinolizidine alkaloids, including matrine, oxymatrine and lupeol. Matrine inhibits cell proliferation [146] (including proliferation of cancer cells)[147][148] Matrine prevents cardiac arrhythmias by inhibiting ouabain-induced calcium overload in guinea pigs.[149] Matrine has neurotoxic properties in Zebrafish (Danio rerio) larvae.[150]
Dissolved oxygen
Fish take up oxygen through the limited gill surface area. At low dissolved oxygen (DO), more metabolic energy is used by the ventilatory system to maintain O2 uptake, which limits weight gain and growth.[151] Feed intake and growth at 5.6 ppm (mg/L) DO is significantly higher than at 3.0 ppm, particularly in big fish.[152] For fish over 200 grams, feed intake continuous to increase over 3.0 ppm DO (with 5.5 ppm being the incipient DO), but digestibility is higher at suboptimal DO.[153] DO levels in ponds increase during the night due to vertical circulation. Organic materials consume oxygen during decomposition.[154] DO levels sharply decline after feeding. Dissolved oxygen levels should be maintained above 5.0 ppm for best growth. At DO levels between 3.0–5.0 ppm feeding should be reduced, and feeding should be stopped at DO levels below 3.0 ppm. Low DO increases the toxicity of ammonia (NH3 unionized ammonia > 2.0 mg/L is lethal for tilapia, growth is inhibited below 1.0 mg/L). [155] Under recirculated water conditions, mean body weight of Oreochromis aureus was higher at 6.5 ppm DO (mg/L), but food conversion ratio was best at 3.7 ppm DO.[156] Intermediate densities of phytoplankton produce higher dissolved oxygen concentrations. Dissolved oxygen levels may be raised by increasing algal growth, not necessarily by reducing algal biomass.[157] DO concentrations at dawn is not related to manure input.[158] Low DO increases mortality due to Streptococcus infection.[159]
Salinity











Oreochromis niloticus may grow better in 7.5 ppt than in 0 ppt salinity [160] and may fairly tolerate salinity up to 10 ppt [161] Tilapia may tolerate 0.5% salinity (5 ppt).[162]
At 8 g / L (8 ppt) salinity adult Oreochromis niloticus may be stocked at 40 fish / m3. At 15 g/L (15 ppt) salinity total production is more than halfed.[163] Hatching success for eggs spawned by yearling females may be similar in freshwater (30.9%), 10 ppt (32.7%) and 15 ppt (36.9%), and considerably higher for eggs spawned at 5 ppt (51.6%). [164] Energy required by Oreochromis niloticus for osmoregulation is the least in the absence of an osmotic gradient (11.6‰ salinity) [165] In freshwater, heating water to temperatures above 27°C is not justifiable, while at 18 or 36 ppt salinity, heating water to 32°C can maximize growth rates without lowering growth efficiency.[166] An increase in salinity inhibits growth, compensated by higher temperatures. Juvenile Oreochromis niloticus may be grown similarly in 28°C and freshwater as in 32°C and 8 ppt salinity.[167]
Oreochromis mossambicus tend to have lower growth rates than Oreochromis niloticus. Oreochromis mossambicus is euryhaline and grows more rapidly in brackish water. (50% seawater)[168] Seawater salinity is 35 ppt. Oreochromis mossambicus reared in salt water grows significantly larger than fish reared in fresh water. [169] This may be due to salt secretion by highly activated chloride cells in branchial and opercular epithelia [170] and/or a decrease in the resting metabolic rate (which decreased with increasing fish size).[171] In freshwater, Oreochromis mossambicus prefers 32°C, and higher temperatures with increasing salinity.[172] Increasing dietary energy concentration increased mortality in Oreochromis mossambicus. The optimum protein to energy ratio for rapid growth, efficient feed conversion and maximum retention of protein and energy appears to be approximately 23.8 mg of protein per kJ energy.[173] Addition of 17α-methyltestosterone enhances its growth beyond that produced by masculinization alone.[174] Of the administered 17α-methyltestosterone, less than 1% remains after 100 hours following withdrawal.[175] Oreochromis mossambicus may cope with either high osmolality (564 mOsm/L), 10 pH or 40°C for several weeks.[176]
Oreochromis mossambicus (♂) × Oreochromis niloticus (♀) may produce 100% male offspring ('red hybrid tilapia') [177](p.363), but spawning frequency may be very low [178] and infiltration of parental broodstock by hybrids may be difficult in mass production.[179] (Thai) Red Tilapia has sexual growth dimorphism in which males grow significantly faster than females. Sex reversal may be induced by fluoxymesterone in 4 days old fry.[180] In 12 ppt salinity, 5 to 12 days old Red Hybrid fry (stocked at 5,660 fish/m3) supplemented with 60 mg 17αethynyltestosterone (ET)/kg diet for at least 7-14 days produces nearly all-male populations (94.3-98.1%).[181] Oreochromis mossambicus (♀) × Oreochromis niloticus (♂) hybrids are relatively salinity tolerant. During 2 months, fingerling growth was not significantly different in 0 ppt, 17 ppt and 32 ppt salinity.[182] Median Lethal Salinity (50% survival in 96 h after transfer from freshwater) ranged from 17.2‰ at 30 days post-hatching to 26.7‰ at 60 days post-hatching.[183] This may be due to Morphological changes in the oesophageal epithelium.[184] The hybrid is superior to Oreochromis mossambicus at all salinities and to Oreochromis niloticus at salinities above 10 ppt.[185] This hybrid may be deprived of food during one week in a 8 weeks period without negatively affecting growth.[186] Due to starvation, the percentage of polyunsaturated fatty acids (PUFA) increases significantly in these hybrids.[187] Growth rate, conversion efficiency and maximum food consumption in Red Hybrid Tilapia may be optimal at 28°C [188] and in 0 to 15 ppt salinity.[189]
Alkalinity & pH
Alkalinity is the "buffering" capacity of water (as containing carbonates and bicarbonates) to resist a change in pH (pH fluctuations). In general, culture waters with pH below 7.0 and total alkalinity below 50 mg /L will get benefits by liming.[190] Liming is the application of high calcium-carbonate (and/or high magnesium-carbonate) rocks to the water. To increase pH, add sodium carbonate (Na2CO3) or calcium carbonate (CaCO3). Sodium carbonate is more effective due to its higher solubility. Sodium carbonate may be extracted (as Barilla) from the ash of plants growing in sodium-rich soils, such as Halogeton sativus (aka Salsola sativa), Salsola soda and Salsola kali. The harvested plants are dried and subsequently burned. From this ash, the (water soluble) sodium carbonate is extracted with water. This solution is boiled dry, resulting in "barilla".[191] Calcium carbonate is the main component in snailshells and eggshells, and present in rocks such as limestone, chalk, marble, travertine and tufa. Limestone (mainly calcite and aragonite) is partially soluble in acid. Calcite water solubility decreases as temperature increases. Coquina is deposited shells (from molluscs etc) containing calcite and phosphate, and may be used as a fertilizer. It is soft and hardens by drying. Chalk is a porous sedimentary calcite rock, formed under water. Travertine forms from geothermal springs. Tufa (mainly calcite, plus some aragonite) is a soft, highly porous variety of limestone. It may mainly form in wetlands, at springs, waterfall cascades (fluvial tufas), and at the periphery of lakes (lacustrine tufa).
To increase alkalinity, one may add sodium bicarbonate, coquina, calcite rocks or limestone rocks, so that the water will dissolve large amounts of calcium and magnesium.[192] In Ghana, coquina may be found in the Akuse region (10 km south-east of Kpong; 53% Calcium carbonates)[193], and along the Gulf of Guinea coast. Large limestone deposits can be found in Nauli (near Axim; >49% calcium carbonate [194]), Buipe, Bongo-Da (Northern; >40% calcium carbonate[195]) and Oterkpalu (near Koforidua; 38% calcium carbonate[196]), and minor deposits at Daboya (Northern), Anyaboni, Dedeso and Asuboni (next to Bawdua) in Eastern, the Ejura scarp (Ejurachem scarp in Ashanti), Kintampo and Prang (Brong Ahafo). [197]. Minor occurrences of limestone, suitable for small-scale production, have been found in the Salaga-Yeji area and the Du-Walewale area in the northern region.[198] To create sodium bicarbonate, sodium carbonate may be dissolved in water and treated with carbon dioxide. Solid sodium bicarbonate will precipitate. To increase the alkalinity of 10.000 L water with 10 ppm, add 168 g. of sodium bicarbonate.[199] Adding sodium carbonate or calcium carbonate decreases the level of free ammonium ions.[200] To decrease water pH, add driftwood, or use peat soil as a liner.
In waters with algae (eg Spirulina), the algae will take up CO2 for photosynthesis. As a result, less CO2 reacts with H2O to form carbonic acid (H2CO3). This turns HCO3- (bicarbonate) and CO3-2 (carbonate) ions into the major forms of inorganic carbon, increasing alkalinity (and DO).[201] CO2 may also be withdrawn (which results in an increase in pH) by adding (bi)carbonates. (Example formula: Na2CO3 + CO2 + H2O => 2NaHCO3)[202]
The appropriate levels for tilapia aquaculture are 20-150 mg CaCO3 / L alkalinity and 7.4-8.2 pH.[203], or 7.5 to 8.5 pH, up to 9 pH.[204] Adding sodium carbonate to water with 20 mg/L CaCO3 alkalinity, increasing alkalinity up to 77 meq/L, does not affect Oreochromis niloticus growth rates. Calcium carbonate is better for tilapia growth, as increasing water hardness as well.[205] Total hardness refers to the content of soluble Ca2+ and Mg2+ in the water. Tilapia need to absorb these ions from the gills for several physiological functions. When total alkalinity is much greater than hardness, water pH may reach 11 in the mid-afternoon due to photosynthesis, because more free CO2-3 remains in solution.[206] Acidifying processes decrease alkalinity, but not hardness of the water.
Fish stocked at higher densities may show better survival rates when total water alkalinity is increased from 30 to 80 or 130 mg CaCO3 per L.[207] Water alkalinity of 35 to 53 mg CaCO3 / L water may be optimum for larval tilapia growth.[208] For actual optimum alkalinity levels for Oreochromis niloticus growth, the total hardness/total alkalinity ratio seems to be key and needs to be greater than 1.[209] Growth of male sex reversed juvenile Oreochromis niloticus is higher at 146 mg/L calciumcarbonate than at 82 mg/L (the difference is actually in calciumcarbonate). A simultaneous increase in water hardness and alkalinity (due to the application of CaCO3) induces better growth than only an increase in alkalinity. The best conditions for Oreochromis niloticus growth are 7.4-8.2 pH; total alkalinity > 50 mg CaCO3 / L; calcium hardness > 140 mg CaCO3/L and free CO2 < 7 mg/L, [210] though free CO2 in water, even when in high concentrations, seems to be harmless to fish when there are suitable concentrations of dissolved oxygen in water.[211] To lower a too high pH, one may add CO2 (or duckweed) to high-DO water.
Oreochromis mossambicus may thrive in water with 245 mg/L bicarbonates [212] or 289 mg/L total alkalinity and 8.8 to 9.4 pH [213][214][215] or 7.9 to 8.6 pH (dominant fork length is 24 cm)[216], may tolerate pH up to 9.6.[217], and may cope with 10 pH for several weeks.[218]
Oreochromis alcalicus grahami (Alcolapia grahami) has adapted to living in waters at pH 10.5.[219], It thrives in Lake Magadi, Kenya (pH 10, 180 mmol CO2/L, 30–40°C)[220] (245 meq/L titratable base, and pH 9.85), where it primarily consumes Arthrospira platensis.[221]
Water flow
A flow rate of 0.5 L/min per kg biomass appears to be desirable for intensive culture of tilapia.[222]
Stocking density
Tilapia needs to be stocked by age group to prevent cannabalism. The risk of cannabalism increases with increasing size differences.[223] Given reasonable assumptions about production costs, the optimal final density of Oreochromis niloticus in a tank-based flow-through system is 73.7 kg/m3.[224] Combined net yield of both caged and open-pond tilapia was highest in the treatment with 50 large fish (av. 141 g.) per m3.[225] Broodstock may be best stocked at 4 fish per m2.[226] Red tilapia stocked in a pulsed-flow aquaculture system at densities of 10 and 20 fish/m3 had significantly higher individual weights than fish stocked at 30, 50, and 70 fish/m3, due to limiting water quality; ammonia concentrations may be high (4 mg/L), and dissolved oxygen low (2 mg/L).[227] Oreochromis niloticus fry may grow optimally when stocked at 5 fry / L and fed at 30% bodyweight / day.[228] Oreochromis niloticus fingerlings may be stocked at a density of 42.6/m3 with a production of 18–20 kg/m3 in 210 days at a water flow rate of 1 L/min/kg fish biomass.[229] With dissolved oxygen levels below 2 ppm (or even below 1 ppm) most of the time, stocking 3 fish per m3 was more profitable due to increased mortality and lower growth rates at higher stocking densities (6 and 9 fish per m2), despite similar water qualities.[230]
Growth
Tilapia nilotica fingerlings grow optimally on diets containing 30-40% protein, 12-15% lipids, and 30-40% digestible carbohydrate.[231] Where extra protein (from 25% to 30%) did not increase growth in young tilapia (av. 18 cm long, 62 g. weight), increasing feeding levels to 2% of bodyweight / day did.[232]
juvenile Oreochromis niloticus grow better at 26 and 30°C, than at 22 and 34°C.[233] Feed efficiency and protein efficiency ratio are better and juvenile fish grow better at 28°C than at 22°C and 34°C. Liver gluconeogenesis and lipogenesis are increased by low temperature (22°C).[234] Growth and feed utilization efficiency is similar in genetically improved farmed tilapia (GIFT), genetically male Nile tilapia (GMNT) and conventional Nile tilapia (CNT).[235] Large Oreochromis niloticus (av. 288 g.) grow less in recirculating aquaculture systems compared to flow-through systems. The opposite is true for small fish (av. 81 g.)[236]
The use of tuna by-product meal as a total replacement for fish meal into tilapia diets increases oxidative stress.[237]
Higher energy conservation is achieved when fish are exposed to longer rather than shorter photoperiod cycles.[238]
If reproduction can be delayed in the females, average growth rates comparable to those of an all-male population may be achieved. [239]
Broodstock
Tilapia reach sexual maturity in 3 to 6 months. A sex ratio of 1:2 to 1:3 (male:female) and a density of 2 fish/m2 gave the best result regarding the number of of seed / female / day and number of seed / kg female / day. Maximum seed production / female / day was recorded for 3-year old females. In tropical regions, monthly fry production may be 10.000 fry per 10 kg female brood. In a broodstock rotation strategy, brood fish are sexed after a spawning cycle and maintained seperately for 10 to 14 days before rotating back to a spawning cage. Broodstock may be tagged through panjet marking (panjeet tattoo); injecting tattoo pigment (Alcian blue stain in solution, or a pink-fluorescent dye / pink photonic dye) into the fin rays with a (dental) jet inoculator (needleless injector; eg dermo-jet, dermojet, syrijet, madajet). About 9 locations are available for marking. By selecting 3 of these locations on each fish, a total of 84 combinations is possible. (the first fish is marked 1,2,3, the second is marked 1,2,4 etc., and the last one is marked 6,7,8.
Incubation
Eggs removed from female mouth-brooders must be incubated. Because the eggs sink, they must be incubated in conical up-welling or down-welling incubators. Stocking rate in 20L incubators is about 4000 eggs per liter. Water flow rate is about 1 L / second per 10.000 eggs. (Little 1989) High quality water saturated with dissolved oxygen is essential in incubators. Survival from eggs to 10 days old fry is usually above 80%.
Strains
Compared to Oreochromis niloticus strains from Egypt and Senegal (bred from wild collected stocks), and ‘Israel’, ‘Singapore’, ‘Taiwan’ and ‘Thailand’ (strains maintained for aquaculture purposes), a strain bred in Ghana from wild collected stocks showed a significantly lower body weight at 210 days. [240]
Triploidy may be induced in Oreochromis niloticus by heat shock. No differences in growth between triploids and diploid controls may be observed at age of maturation, but triploids are significant heavier (P < 0.01) than control fish at the end of the experiment. Triploid males exceeded body weights of respective controls on average by 66 ± 17%, whereas triploid females displayed an even higher increase in body weight compared to diploid ones (95 ± 27%).[241]
Masculinization
Estrogen synthesis is crucial for ovarian differentiation, and transcription of the aromatase gene can be proposed as a key step in that process in fish. Treatments with an aromatase inhibitor (ATD, 1,4,6- androstatriene-3-17-dione) result in 75.3% masculinization of an all-female (XX) population in tilapia (dosage 150 mg/kg of food). The effectiveness of the aromatase inhibition by ATD is demonstrated by the marked decrease of the gonadal aromatase activity in treated animals versus control.[242]
Aromatase activity as a key factor in sexual differentiation in Oreochromis niloticus.[243] The most sensitive time to aromatase inhibitors lies in the first week (between 7 and 14 days post hatch). Treatment with the aromatase inhibitor Fadrozole (nonsteroidal) showed a dose-dependent increase in the percentage of males from 0 to 200 mg per kg. At higher doses (200 to 500 mg / kg), the percentage of males remained more or less constant (92.5-96.0%).[244]
The masculinizing actions of 17alpha-methyltestosterone (MT) are most potent at up to day 20 of age.[245] Treating female tilapia Oreochromis niloticus with methyltestosterone (at a dose of 50 mcg/g diet) resulted in 100% masculinization.[246] Fry may be fed a commercial diet (SRT-95) with 30 ppm α-methyltestosterone for 21 days to yield 99% males.[247] Numerous papers have reported that adding 17α-methyltestosterone at 60 mg/kg to the diet for several weeks induces mono-sex male populations. During growing-out season, muscle testosterone concentration in monosex tilapia was sharply decreased to 2.625 ± 0.303 ng/g at the harvesting date. [248]
Treatment with tamoxifen and letrozole (200mg/kg feed) to fingerlings of O. niloticus for 60 days brought about 98.5% masculinization. Treatment with 17α methyltestosterone (35 mg/kg feed) to fingerlings of O. mossambicus after 8 days post hatch for 60 days obtained 100% sex reversed males with excellent growth.[249]
During the restricted developmental period temperature is of great influence. The critical period for elevated-temperature-induced masculinization lies between days 10 and 15 post-hatch.[250] Higher temperatures (during 5 days) before they are 5 days old induces deformities. Masculinization is induced at elevated temperatures (28 to 32°C) during 5 days after 10 days old.[251] The advantage of producing faster growing males of Nile tilapia at high temperature would hardly compensate the loss of production incurred during the masculinising treatment.[252] High-temperature (almost 36.90 °C) treatments yield a significantly higher proportion of males (64.20–80%) with lower survival rates (60–81%). The sex ratio of progenies reared at temperature below 36°C never deviated significantly from the balanced sex ratio.[253]
Feminization
The critical period for low-temperature-induced feminization lies between days 5 and 10 post-hatch.[254] The period of maximal feminizing action of 17beta-estradiol (E(2)) upon sex ratio is before 10 days posthatching in tilapia (Oreochromis mossambicus).[255]
During the restricted developmental period temperature is of great influence. Higher temperatures (during 5 days) before they are 5 days old induces deformities. Gonadal feminization is induced at lower temperatures (20°C) for 5 days before 10 days old.[256]
Genetic Masculinization
One may produce genetically male tilapia by creating yy (male or female) broodstock, from 4 unrelated Oreochromis niloticus families: A, B, C and D. After that, you need to add one more family (E, F, etc.) per 3 years. First, one must create both xx and xy females through feminization, and then select the xy females based on the sexe ratio of their offspring.
Phase 1
- 5X: A @ B => estrogen treated => 50% female ABxx + 50% female ABxy (~1500 females)
- After 7 months, 20 AB females are selected for progeny testing with C males: AB @ C (in total 6000 fish per month; 62% male progeny on average) :
- ABxx @ Cxy = 50% males (after sexe outcome, 7 months later, the ABxx females are eliminated)
- female ABxy @ Cxy = 25% ABCxx + 25% ABCyy + 50% ABCxy (75% males)
Phase 2 ; The remaining ABxy females are now broodstock. In the next step, (also) yy females are created, and selected.
- 5X: female ABxy @ Cxy => estrogen treated => 25% ABCxx + 25% ABCyy + 50% ABCxy (~1500 females)
- 7 months of progeny testing of 20 ABC females with D males: ABC @ D (in total 6000 fish per month; 75% male progeny on average) :
- ABCxx @ Dxy = 50% males (after sexe outcome, the ABCxx females are eliminated)
- female ABCxy @ Dxy = 25% ABCDxx + 25% ABCDyy + 50% ABCxy (75% males. After sexe outcome, the ABCxy females are eliminated)
- female ABCyy @ Dxy = 50% ABCDxy + 50% ABCDyy (100% males)
Phase 3 ; The remaining ABCyy females are now broodstock, producing all male offspring. But there are just a few (~33% of 20), so that the previous step is repeated.
- 5X: female ABCyy @ Dxy => estrogen treated => 50% ABCDxy + 50% ABCDyy (~1500 females)
- 7 months of progeny testing of 40 ABCD females with E males: ABCD @ E (87% male progeny on average):
- female ABCDxy @ Exy = 25% ABCDExx + 25% ABCDEyy + 50% ABCxy (75% males. After sexe outcome, the ABCDxy females are eliminated)
- female ABCDyy @ Exy = 50% ABCDExy + 50% ABCDEyy (100% males)
As a result, about 20 female ABCDEyy are now broodstock, and need to be replaced every 3 years, starting with progeny testing 7 months earlier (or 14 months earlier, if maximally 20 females can be progeny tested during the same period). Equally so, the parallel yield of 100% males in phase 2 from different families, will consist of 50% FGHIJxy and 50% FGHIJyy. From this 100% male population, one may select the yy males through progeny testing, as they will produce 100% male offspring. ABCDEyy @ FGHIJyy will create yy broodstock, which consequently will always produce 100% male offspring.
Parasites
Tilapia may be infected with:
- Nematodes / roundworms, such as Contracaecum sp.[257] (eg Contracaecum multipapillatum [258]), Paracamallanus cyathopharynx, Procamallanus laevionchus [259], Rhabdochona esseniae [260], Rhabdochona paski [261], Gnathostoma spinigerum, Gnathostoma doloresi and Gnathostoma hispidum.[262] Human gnathostomiasis infection is related to ingestion of "ceviche".[263]
- Cestodes (aka tapeworms), such as Amirthalingamia sp. (eg Amirthalingamia macracantha; higher prevalence and intensity in wet season [264]), and Cyclustera sp.[265] (eg Cyclustera magna [266])
- Trematodes / flukes (encysted metacercariae)[267], such as Clonorchis sinensis (human liver trematode)[268], Tylodelphys sp.[269], Stictodora tridactyla (in brackish-water fish)[270], Ribeiroia marini [271], Clinostomum sp.[272] ((eg Clinostomum Complanatum, Clinostomum tilapiae, Euclinostomum hetereostomum [273]), Diplostomum sp.[274], Neascus sp, Acanthostomum sp.[275], Bolbophorus sp.[276], Haplorchis yokogawai [277], Haplorchis pumilio, Haplorchis taichui, Centrocestus formosanus, Stellantchas musfalcatus, Echinochasmus japonicus [278], Heterophyes heterophyes, Heterophyes aequalis, Pygidiopsis genata, Stictodora sp., Phagicola sp.[279] (eg Phagicola ascolonga[280]) and Prohemostomumn vivax.[281] These trematodes loose their viability after 48 hours at -5°C.[282] The conditions of gas supersaturation (gas pressure > 111.2% saturation; N supersaturation, O2 undersaturation) may result in heavy monogenetic trematode infections in tilapia.[283] In Thailand, fish-borne zoonotic trematode (FZT) metacercarial infections were found in Nile tilapia from cage (2.5%) and pond aquaculture systems (10%) and in wild caught fish.(53%) [284] In China, tilapia from nursery and grow-out ponds were sampled from monoculture, polyculture and integrated aquaculture systems, revealing a 1.5% prevalence of FZT infections (Heterophyidae and Echinostomatidae); lower than in wild caught fish. Integrated systems using animal manure and latrine wastes as fertilizer did not show a higher prevalence.[285] In Vietnam, the overall FZT prevalence in tilapia from wastewater ponds was 2.0%, but much higher in tilapia from farm ponds. [286]
- Monogenea (very small flatworms), such as Dactylogyrus minutus [287], Neobenedenia melleni [288][289], Enterogyrus cichlidarum (associated with overcrowding [290] and infection intensity increases with (tilapia) host size[291]), Gyrodactylus malalai (ectoparasite)[292], Gyrodactylus ergensi [293], Scutogyrus longicornis [294] and of the genus Cichlidogyrus [295], such as Cichlidogyrus tilapiae, Cichlidogyrus sclerosus [296], Cichlidogyrus dossoui [297], Cichlidogyrus halli [[298], Cichlidogyrus thurstonae [299], Cichlidogyrus berradae, Cichlidogyrus revesati, Cichlidogyrus legendrei and Cichlidogyrus lemoallei.[300] The highest parasite number was recorded in reservoir-dwelling, and lowest in stream-dwelling tilapia.[301] Gyrodactylus niloticus infection may particularly become lethal (in tilapia) when followed by exposure to the bacterial pathogen Streptococcus iniae.[302] Low stocking density and low water temperature may result in lower monogenea parasitism rate.[303]
- Acanthocephala (thorny-headed worms), such as Acanthogyrus (Acanthosentis) tilapiae.[304] and Polyacanthorhynchus kenyensis [305] In Kenya, infection rates in wild Tilapia zillii ranged from 4.1 to 77.7%.[306]
- Ciliatea ('small flagella'), such as Epistylis sp.[307], Paratrichodina africana [308], Trichodina compacta [309], Trichodina magna [[310] and Ichthyophthirius multifiliis.[311] Ichthyophthirius multifiliis load increases susceptibility and mortality of tilapia to Streptococcus iniae (Gram-positive bacteria) infection.[312]
- Dinophyceae (Dinoflagellata; "red tide"), such as Piscinoodinium pillulare.[313] (a dominant parasite in Brasilian tilapia ponds)[314]
- Myxosporea, such as Henneguya suprabranchiae [315], Myxobolus heterosporus [316] (induces total destruction of the intestine structure [317]), Myxobolus nounensis [318], Myxobolus dossoui (host size effect), Myxobolus zillii (seasonal pattern) [319], Myxobolus microcapsularis [320], Myxobolus agolus, Myxobolus brachysporus, Myxobolus clarii, Myxobolus cichlidarum, Myxobolus tilapiae and Myxobolus camerounensis.[321]
- Conoidasida (unicellular), such as Goussia cichlidarum.[322]
- Phyllopharyngea (single-cell parasites), such as Chilodonella hexasticha.[323]
- Trhypochthoniellus longisetus longisetus, a mite.[324]
- Lamproglena sp. (Lernaeidae; small crustaceans) [325]
- Pentasomida (tongue worms; tiny crustaceans), such as Leiperia cincinnalis [326], Subtriquetra wedli [327] and Subtriquetra riley.[328]
- Anodontites trapesialis (the larval stage of this mussel).[329]
No parasite vaccines exist.[330] Tilapia are relatively resistant to parasitic infections.[331] The prevalence of heterophyid encysted metacercariae infection decreases as fish size increases.[332] When dissolved oxygen, temperature, pH, nitrite and ammonia are within normal rate parasites may cause little harm to tilapia.[333] Parasitic infection by Acanthogyrus tilapiae provokes an aggressive host response.[334] Tilapia may produce an induced humoral immune response against Cichlidogyrus sp.[335] Oreochromis mossambicus initiated a strong immune protection by direct exposure with even a small number of parasites.[336] Gyrodactylus numbers fluctuate independently of temperature. In anticipation of immune defenses reaching the fish surface through mucus, Gyrodactylids (Monogenea) worms progressively move away from fins with high mucus cell density to those with low density.[337] Tilapia may also develop resistance to Neobenedenia sp. (Monogenea) infection.[338]
Freshwater mollusks in tilapia ponds, such as Melania tuberculata, Melanoides turricula, Pomacea flagellata, Haitia cubensis and Anodontiles luteola, may host various parasites that are harmful to tilapia.[339] The Biomphalaria sp. snails are often found in tilapia ponds (fresh water reservoirs are their natural habitat[340]) and are intermediates for Schistosoma mansoni.[341] Biomphalaria cf. havanensis is an intermediate host for Diplostomum ompactum, causing higher levels of infections in cultured tilapia than wild tilapia.[342] The snail Bulinus truncatus is an intermediate host for Clinostomum tilapiae, Euclinostomum heterostomum, Bolbophorus levantinus and Neascus-type metacercariae. The snail Melanoides tuberculata is an intermediate host for Centrocestus sp. and Haplorchis sp. The snail Melanopsis costata is an intermediate host for Pygidiopsis genata, Phagicola longa.[343] The snail Thiara granifera is commonly infected with Haplorchis pumilio.[344] Dogs, cats and pigs may also be intermediate hosts for metacercariae.[345]
Author
Author of this article is Thijs Klompmaker, born in 1966
- Author's website, as personal trainer (specialized in isolation exercises and dietary solutions)
- Author's Instagram account
- Author's YouTube Channel